How to Optimize Dehydration Units
Molecular Sieve Process Optimization
Featured in Biofuels International, May 2015 (volume 9, issue 3)
Molecular sieve dehydration of ethanol uses a specialized molecular sieve to remove the water from the distillation/rectification of ethanol and water. Distillation can only remove water to approximately 5% of the total composition.
Molecular sieve consists of tiny zeolite crystals made of sodium aluminum silicate blended in a clay binder and formed into spherical ceramic beads. The crystals are hollow and have three angstrom openings that allow water to be adsorbed under pressure, within the crystal, while the ethanol passes through the molecular sieve to the product stream. The adsorbed water is removed under vacuum and recycled to process water.Before understanding the optimization process, it is important to keep these two general principles regarding adsorption in mind:
- Capacity increases as pressure increases.

- Capacity decrease as temperature increases.
With regard to understanding how we optimize a process, additional principles need to be considered; it is necessary to explain the nature and function of a water adsorption isotherm to fully realize these principles. An isotherm shows the adsorption capacity of the molecular sieve as a function of pressure at constant temperature.
Optimization Adsorption Principle One
Ideal temperature and pressure for maximum working capacity
Based upon the two general principles of adsorption, the obvious conclusion is that we need to operate the unit at the highest pressure and lowest temperature possible. When choosing a temperature for the operation, it is essential to recognize that this is strictly a vapor phase process, meaning the feed stream cannot change phases at any point; thus, the lowest temperature that can be utilized would lie just above the point of a phase change. The system is limited by the maximum pressure sustainable by the system and the amount of super heat available.
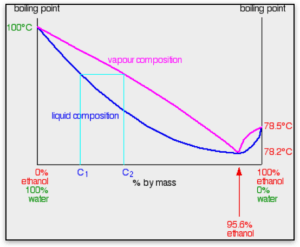
The highest pressure is defined by the ratings of the vessels, piping and valves (consult your design constructor or your P&ID’s to determine system limitations). The lowest temperature is defined by the boiling point of the ethanol/water mixture. Most designs are built around the water/ethanol azeotrope, approximately 95% ethanol (190 proof). In actual practice, most facilities run somewhere below the azeotrope, as low as 90% ethanol (180 proof). Whatever the mixture, the actual boiling point must be determined. In order to ensure the material stays in the vapor phase, it is suggested that the inlet vapor temperature be set at 50°F or 10°C of superheat.
There are a number of boiling point calculators available for free on the internet. Input the composition and the pressure to calculate the boiling point. Your vaporizer must have adequate heat to maintain the excess superheat point of the mixture but not so much excess heat to adversely affect water capacity (remember capacity is inversely proportional to temperature), and excess enough to ensure the mixture stays in the vapor phase but not anymore excess than is necessary.
Determining these variables help pinpoint the ideal conditions for maximum adsorption.
Apply the same isotherm principle to determine the maximum conditions for regeneration. At constant temperature, you need to achieve the maximum vacuum available in the vessel. The difference in the capacity at constant temperature between the highest pressure practically obtainable and the lowest pressure possibly obtainable defines the working capacity.
Optimization Principle Two: Working Capacity
All molecular sieves come with a specification sheet, which should state static water capacity. This has little to do with the actual working capacity as all bound 3A sieve beads have a static capacity between 18-22% water by weight – For example, Hengye Inc., EthaDry 4×8 mesh beads have a static water adsorption of ≥21.0.
Static capacity is important as an overall indicator of the purity of the molecular sieve, but working capacity is most important for operation. Working capacity is defined as the difference in the water capacity of the molecular sieve at constant temperature between the two operating pressures, adsorption and regeneration.
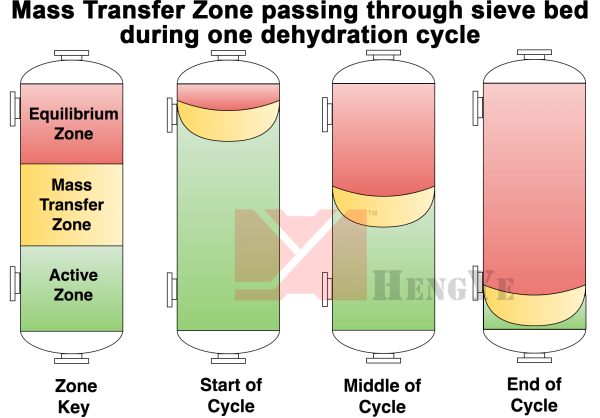
The mass transfer zone is the area during an adsorption or regeneration portion of the cycle where the water is actively being adsorbed or desorbed from the molecular sieve. In theory, the shape of the mass transfer zone is “plug flow,” a cylindrical wafer that uniformly moves through the bed, across the diameter. In reality, the mass transfer zone is shaped by both the vapor distribution and the drag on the walls of the vessel. Proper distribution is required for optimal vapor distribution to ensure full usage of the entire molecular sieve product.
Factors Affecting Working Capacity
- Temperature should always ensure vapor phase maintenance under the given composition and pressure. Two phase flow is detrimental because the liquid forms a barrier on the bead surface, which inhibits mass transfer of the liquid into the crystal interface. Surface tension of the liquid makes it difficult for the liquid water to be removed by re-vaporization once it’s condensed to the liquid phase.
- Distribution of vapors on the inlet. Mass transfer zone – shape and velocity.
At a given temperature, maximize the difference between the adsorption pressure and regeneration vacuum. - Choose the right 3A molecular sieve.
- Compare not only specification sheets, but request sample Certificates of Analysis. Compare molecular sieve products from multiple manufacturers; they are not all created equally and therefore do not perform equally, for example:
a. Crush strength
– A higher number indicates a more durable product
b. Attrition
– the tendency to dust – A lower number indicates a more durable product. Dust in your vessels leads to inefficiency
c. Particle size distribution
– The narrower a size, the better the flow characteristics in the vessel
d. Ethanol co-adsorption
– You want the working capacity dedicated to water not ethanol. The higher the co-adsorption, the higher your regeneration proof. If ethanol co-adsorption is not listed, look at CO2 adsorption as this is indicative of ethanol adsorption.
e. Ask for working capacity (WC), this varies with process, but it’s still important to ask. To calculate WC, figure the mass balance around an actual production unit.
f. Talk to your vendor and tap their technical expertise
– You may have one unit, but they may have filled hundreds.
Understanding the process within your vessels, controlling your parameters, and understanding molecular sieve and how it works, are critical considerations for maximizing the use of your equipment and energy.
Your D&E companies, service companies, and especially your molecular sieve vendor have the expertise to help you understand the process and get the most out of your ethanol dehydration unit. Not all molecular sieves and molecular sieve vendors are the same.
Authored By:
Mark Binns
Hengye Inc.
Houston, Texas
info@hengyeinc.com
1-844-308-3271
A slideshow format about Process Optimization is also available: Optimizing Dehydration Units (Vapor Phase)
DISCLAIMER NOTICE
The above information is for general reference and guidance purposes only, Hengye does not accept any responsibility for actions based on the information above. Each reader/operator must consult equipment/operation manuals, the plant engineering/design company, and the plant/equipment builder before performing any work.


