The Role of Co-adsorption
Molecular Sieve Manufacturing
All Type A molecular sieve originates as Type 4A zeolite crystals. These 4A zeolite crystals can be treated to an ion exchange to create 3A or 5A molecular sieve. To create Type 3A zeolite, larger potassium ions exchange for smaller sodium ions within the crystal matrix, thus shrinking the pore opening of 4A molecular sieve (measuring approximately 4 Angstroms in diameter) and creating Type 3A molecular sieve, which pore size measures about 3 Angstroms in diameter. For Type 5A, a smaller calcium ion is exchanged with a sodium ion in the crystalline structure to create a larger pore aperture, making a less selective zeolite compared to Type 4A. A perfect and total ion exchange, however, is not feasible, so a certain amount of molecular sieve crystals will retain a 4A structure. For Type 5A molecular sieve, having residual 4A zeolite does not have a major impact, but for Type 3A molecular sieve, the residual 4A crystals can cause the undesirable co-adsorption of molecules that measure between 3 and 4 Angstroms.
The Significance of Pore Size
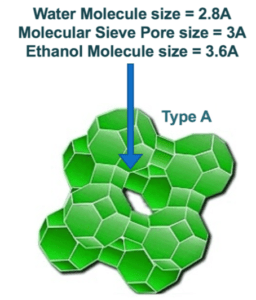
When comparing the actual performance of various Type 3A molecular sieves, it is important to consider the co-adsorption rate. For processes such as Ethanol Production, co-adsorption is an undesirable occurrence and refers to the amount of ethanol that is adsorbed by the molecular sieve along with the water that is intended to be adsorbed. Ethanol is not adsorbed by 3A zeolite because the pore opening of 3A molecular sieve crystals is too small to adsorb ethanol molecules as these molecules measure approximately 3.6 Angstroms in diameter. Water molecules only measure about 2.8 Angstroms in diameter, so the 3 Angstrom pore aperture of Type 3A is ideal for adsorbing water and excluding ethanol, allowing ethanol to pass through to the product stream. Given the diameter of both water and ethanol molecules, a 4A crystal, measuring approximately 4 Angstroms in diameter can adsorb both water and ethanol, so while water is adsorbed, some ethanol will be adsorbed as well.
How Can One Sieve Differ from Another?
The degree of co-adsorption varies by product selection and by manufacturer, based on the efficiency of the ion exchange rate or theoretical quality of 3A molecular sieve. Some manufacturers use specialized methods to exchange ions and reduce 4A crystals to a minimum presence, which decreases the co-adsorption of ethanol and other larger molecules during the dehydration process. While the ion exchange rate is imperative to co-adsorption, the crystallinity, clay binder choice, macro porosity, and other factors also play a role in 3A co-adsorption properties.
Lab Testing Methods to Determine Co-adsorption Properties
Molecular sieve can be tested and compared both in laboratory settings or in real life scenarios to determine the impact that co-adsorption plays on production efficiency. In a lab, the purity of 3A molecular sieve can be determined by comparing the heat of adsorption of a sample exposed to pure water to a sample that is exposed to various molecules such as ethanol, methanol, carbon dioxide, and more. For this example, we will be comparing water adsorption to ethanol adsorption. The heat of adsorption is measured by a thermometer placed in a beaker of molecular sieve. Water is added to the beaker and the change in temperature is measured. Similarly, a thermometer is placed in beaker of molecular sieve and ethanol that is 199 proof or 99.5% purity or greater is poured into the beaker and the change in temperature is measured. The changes in temperature are compared and indicate the amount of 4A molecular sieve that is present in the molecular sieve sample. Molecular sieve adsorption is an exothermic reaction that releases heat during the adsorption process. So, if a 3A molecular sieve shows a large temperature change when exposed to nearly pure ethanol, there is an indication that a large amount of 4A molecular sieve present, which is adsorbing ethanol and creating a heat increase caused by the adsorption process. A sample with lower compositions of 4A crystals will have a lower change in temperature when exposed to nearly pure ethanol, so a small heat increase indicates that there is a low composition of 4A crystals in the molecular sieve beads.
Testing in an Operational Ethanol Plant
In real life scenarios, operation data can be used to compare feed rate with product feed rate. Feed rate into the system is used to determine the amount of water and ethanol coming into the dehydration beds while product feed rate can measure the amount of ethanol coming out of the system. The ethanol that is not released into the product stream should appear in the regeneration feed to the condenser and can indicate the amount of ethanol being recycled through the system.
Dehydrating Ethanol
In Ethanol Production, one of the last, essential production steps is to dehydrate the ethanol to about a ninety-nine percent purity. Achieving such high levels of ethyl-alcohol purity cannot be accomplished by distillation alone because a natural phenomenon occurs, called an azeotrope, which prevents water from further being boiled out of the mixture. This azeotrope forms at about a ninety-four percent ethanol purity, and ethanol this low in purity is not generally considered suitable for use as fuel or as a fuel additive. To further dehydrate the ethanol, Type 3A molecular sieve is applied to the dehydration process. This molecular sieve will selectively remove water most of the remaining water, to create about ninety-nine percent pure ethanol, which will pass through the dehydration beds and along to the product stream.
The Impact of Co-adsorption in Ethanol Production
Co-adsorption of ethanol is a problem for ethanol producers because capacity for water per cycle is decreased when ethanol is adsorbed, which leads to shorter cycle times and lesser volumes of ethanol being produced during the given cycle. Co-adsorption of ethanol also means the regeneration proof will be higher than necessary, which means that more ethanol will be recycled and returned to the rectifier, ultimately decreasing the production capacity per cycle while simultaneously losing efficiency and capacity for future dehydration cycles. This occurrence leads to the reprocessing of the same ethanol repeatedly, instead of sending the ethanol out as dry product. In practice, the purpose of molecular sieve is to remove water from ethanol, and not to recycle ethanol throughout the system. The occurrence of ethanol recycling costs operations money through loss of product, repeated expense of energy, and reduced working capacity of the dehydration units. Although the working capacity and efficiency may only equate to a few gallons per minute, these savings extrapolated to the course of 350 operation days per year can quickly add up to over one million gallons per year. This difference in capacity should be focused to become and increase instead of a loss in production, which will increase the plant’s overall profitability. It is not recommended to choose sieve suppliers based on immediate price alone, ethanol co-adsorption should be deeply considered, along with other factors such as crush strength and adsorption capacity.
Authored By:
Mark Binns and Kolten Burkes
Hengye Inc.
Houston, Texas
info@hengyeinc.com
832.288.4288


